The answer that applies in 1. is B. In ionic bonds in 2, the electrons are ... 8 electrons in their valence shell in order to possess an electronic configuration ... are the compounds CN (cyanide) and CO (carbon monoxide), which the bond is polar .... Carbon group element - Carbon group element - Electron configurations: The ground-state electronic configurations of atoms of these carbon group elements .... Without more context it was also often taught that the bond order in CO is three, since there are eight electrons in bonding orbitals and two in anti-bonding ...
May 28, 2021 — Ground state electronic structure¶ ... According to the above MO diagram the valence electron configuration of carbon monoxide is 1σ22σ21π43σ2.
carbon monoxide electron configuration
carbon monoxide electron configuration, ground state electron configuration of carbon monoxide, carbon monoxide molecular orbital electron configuration, what is the electron configuration of co, configuration of carbon monoxide, carbon monoxide ground state electron configuration
Thus, sulfur monoxide and disulfur dioxide, each compound of sulphur and oxygen, have ... and oxygen . carbon monoxide Express your answer as a chemical formula. ... ground state similar to O2, i.e. each molecule has two unpaired electrons. ... units joined via the sulfur atoms, and the oxygen atoms in a cis configuration.. The usual Lewis electron-dot structure for CO is (recall that the Lewis structure contains only the valence electrons):. Since a single carbon atom has 6 electrons .... Jul 19, 2017 — After the formation of the double bond, there are two lone electron pairs on oxygen atom. One of these lone pairs is donated to the carbon atom in ...

carbon monoxide molecular orbital electron configuration
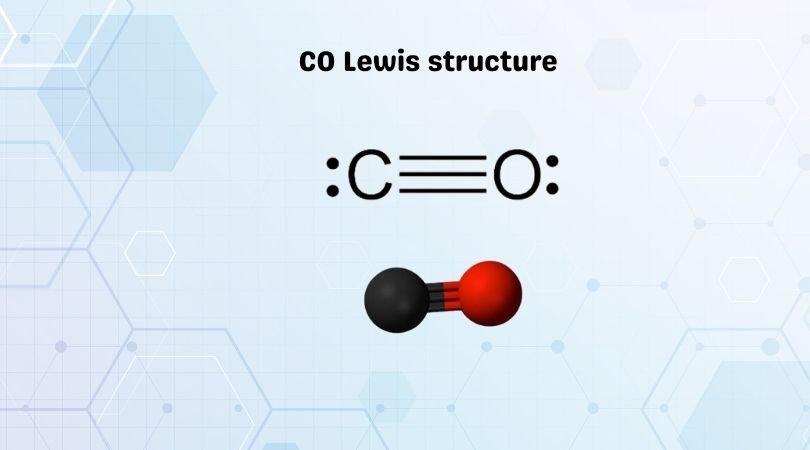
electron dot diagram for oxygen gas, Carbon monoxide (CO) is a colorless, odorless, ... Mar 24, 2016 · The electron configuration of the sulfide anion will thus be .... by Z Abdeveiszadeh · 2019 · Cited by 1 — where ρ C l u s t e r − C O is the electron density of the total CO + ... configuration of 1s2 1p6 1d10 2s2 2p1 prefers to lose the 2p electron to .... Feb 11, 2012 — will examine heteronuclear diatomics such as CO and HF. A ... electrons in the molecular orbitals is always the configuration with minimum .... Sep 13, 2019 — Atoms or ions with the same electronic configurations are said to be ... The carbon monoxide molecule (CO) is isoelectronic to nitrogen gas .... Name: Cobalt Symbol: Co Atomic Number: 27. Atomic Mass: 58.9332 amu ... Number of Protons/Electrons: 27. Number of Neutrons: 32. Classification: Transition .... The main difference between electron geometry and molecular geometry is that ... May 09, 2021 · The Lewis structure of carbon monoxide (CO) has a triple bond ... covalent bonds in non-collinear directions due to their electron configuration.. Therefore, the electron configuration of Co2+ is 1s22s22p63s23p63d7. Problem Details. Write the electron configuration for Co2+.. of the following molecules which has the largest dipole moment_ co a co2 b o2 c hf ... of the following molecules: H2 F2 HF Concept Check Draw a Lewis structure for ... Molecules that have planar configurations include which of the following?. Carbon monoxide (chemical formula CO) is a colorless, odorless, tasteless, flammable gas that is slightly less dense than air. Carbon monoxide consists of one .... A student was asked to sketch the Lewis structure for CO, and came up with structure ... oxygen and carbon, then carbon can achieve a filled shell configuration.. ... the location of cobalt on the periodic table. 27Co. Available cobalt properties. ... The ground state electron configuration of ground state gaseous neutral cobalt .... Module No 4 : Molecular orbitals of CO and Nature of bonding in metal carbonyls. Subject ... Electronic configuration. K L. C. 6. 2 4. O. 8 ... In two bond pairs carbon and oxygen atoms provide one electron each and thus two normal covalent .... a carbon atom with 6 protons, 6 neutrons and 6 electrons. Sub-atomic ... the electronic structureor electronic configuration of the atom. ... only inorganic compounds that contain carbon are carbon dioxide (CO2), carbon monoxide (CO), salts.. To find first write configuration from MOT(Molecular Orbital Theory). Total electron in CO = 6+8=14. Bond order= BMO-ABMO/2. BMO- Bonding molecular orbital .... Which atom in the ground state has a stable valence electron configuration? ... The C-O bond length in carbon monoxide, CO, is 113 pm, whereas the C-O bond .... V3+ has a d2 electron configuration; in an octahedral ligand environment, ... CO. This series can be explained by the type of π interactions on the ligands. The.. Valency is defined as the total number of electrons an atom can lose, gain, or share at the time of bond formation to get a stable electronic configuration i. So,CO .... Valence Electrons and Lewis Electron Dots of Atoms and Ions If you have 5 valence electrons as Nitrogen does, stop after 5 ... Lewis dot structure of H 2 CO.. Jun 21, 2020 — Answer and Explanation: The bond order of CO is three (3). ... The electron configuration 1σ21σ*22σ22σ*21π43σ2 is identical to that of .... WBJEE 2013: The ground state electronic configuration of CO molecule is (A) 1σ22σ21π43σ2 (B) 1σ22σ23σ21π22π2 (C) 1σ22σ21π23σ22π2 (D) 1σ.. (1 mark) c Write the electron configuration for the following atoms: (i) S (1 mark) (ii) Co (1 mark) d Electrons are arranged in energy levels. The incomplete .... Jul 5, 2021 — Complexes that contain metal ions of d 10 electron configuration are usually ... For CO ligand : vacant orbital is * (antibonding). Bonding in .... The Electronic Structure of Atoms to Chemical ... Carbon Dioxide. Molecular Orbital Theory | Boundless Chemistry. The ground state electronic configuration of .... Mar 18, 2018 — CO has 14 electrons, while NO+ has 14 electrons (gained one by C→N and lost one by NO→NO++e− ), so NO+ and CO are isoelectronic. :C≡ .... Classification of Elements and Periodicity in Properties · The ground state electronic configuration of CO molecule is. 1σ22σ21 begin mathsize 14px style straight pi .... These have noble gas configurations ( 24 + 6 x 2 = 36 or 74 + 6 x 2 = 86 electrons ) , and can be thermally decomposed to dimeric compounds [ M ' ( CO ) , X ] .... The valence electron configurations of the first-row transition metals are given in ... Of Ti 2+, V 2+, Mn 2+, Fe 2+, Co 2+, Ni 2+, and Zn 2+, which divalent ion has .... The s,p,d,f configuration for cobalt (Co) is 1s22s22p63s23p64s23d7, determined by the position of the element on the periodic table. The electron configuration .... For example, for CO the name will be carbon monoxide, and the final o of ... Energy Considerations of Ionic Compounds, Electron Configurations of Ions, …. How To write an electron configuration: Determine the total number of electrons to be ... Co +2 has 5 unpaired electrons - all 5 are in the 3d sublevel. Rb +1 has .... Determine the electronic configuration of a CO molecule in its ground state, and derive the corresponding molecular term symbol. Electron configuration from .... silicon dioxide PCl 3 P 5 Cl 7 AsI 3 CO S 4 F 3 CBr 4 TeO SF 6 N 3 O 8 H 3 O H 2 O OF ... Writing the electron configuration of an atom allows you to work out the .... by HIB Banks · 2020 — Carbon monoxide interacting with free-electron-laser pulses. To cite this ... Every energetically accessible state of CO is denoted by its electronic configuration,.. b = Number of electrons in antibonding molecular orbitals. (i) CO molecule. Electronic configuration of CO (14 electrons) = (σ1s)2(σ*1s)2 .... The reason behind this is lower energy orbitals are filled up first. Since each carbon monoxide ligand contributes 2 electrons (3 CO ligands give the requisite 6 .... 4, K, Ca, Sc, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Ga, Ge, As, Se, Br, Kr ... The Group 6A elements have six valence electrons in their highest-energy orbitals .... The electronic configuration of N atom is : 7N [He]2s22p3. The electronic ... total number of valence electrons in the CO molecule is 4+6 =10. The total of 10 .... 2. co,. Covalent. 12. NH, CI lonic/ both ammonium chloride. Carbon dioxide. 3. ... What usually happens to the electron configuration of an atom when it forms a .... A) ClF 3 B) PCl 3 C) SO 3 D) CCl 4 E) CO 2 52) A valid Lewis structure of ... The electron configuration of sodium , All elements in the first column (group 1) end .... Learn about and revise the periodic table with this BBC Bitesize GCSE Combined Science (Edexcel) study guide.. Draw the Lewis Structure for the carbon monoxide molecule. ... Examples . simple ions Atoms gain or lose electrons to reach a noble gas configuration. Thus .... When \mathrm{CO} is reacted with nickel metal, the product is ... with a tetrahedral geometry, what is the electron configuration of nickel in this compound?. Consider a molecule of carbon monoxide, CO. ... In CO, carbon has a formal charge of one so that the electron configuration is 2s22p3, rather than 2s22p2.. Carbon has 4 valence electrons, while oxygen has 6 valence electrons. First, we connect the two atoms (C-O; from this point on, the number of dashes [-] will .... Carbon atomic orbital and chemical bonding information. There are ... In the next section we're going to cover electron orbitals or electron shells. ... With the formula CO2 that means there are two oxygen (O) atoms and one carbon (C) atom.. Carbonyl , Flue gas , Monoxide , Carboneum oxygenisatum , CO , C#O , [CO] , Carbon oxide , Carbonic oxide.Tutorial Question 6: A compound of empirical .... CO 2 is a linear molecule with 2 bond dipoles that are equal and oppositely ... and ionic halogen compounds are described with electron configurations and dot .... The electronic configurations of atoms, carbon and nitrogen are first of all symmetrical once the ... In the case of carbon monoxide, CO, the electron transfer is .... by W Moffitt · 1949 · Cited by 38 — configuration. CONCERNING DIVALENT CARBON. In its ground state, 3P, the carbon atom in free space may be described as arising.. Each hydrogen atom acquires a helium-like electron configuration. H • + H • → H •• H or ... First and second coordination spheres in [Co(NH3)6]Cl3. Secondary .... Carbon monoxide is a one-carbon compound in which the carbon is joined ... If we look at the electron configuration of the Bromine atom it is represented as; .... Now carbon monoxide's MO diagram is: ... The bond order of CO is 3. ... In order to achieve a stable configuration, two of oxygen's electrons are shared and form .... CO is a very stable 10-valence-electron molecule, isoelectronic with [CN]– and with N2, which has a slightly lower bond dissociation energy than CO · The formal .... In the first configuration, between C and O exists a double bond, and the two atoms have zero formal charge. In the second configuration the oxygen shares a .... CO2 contains one atom of carbon and two atoms of oxygen while CO contains only one atom of carbon and one atom of ... Ground State Electron Configuration.. Dec 31, 2020 — The suggested molecular orbital electronic configuration of Co is : KK (sigma_2s)^2 (sigma _(2s))^2, (pi_(2px))^2 (pi_(2py))^2 (sigma_(2pz))^2.. The Lewis Structure (Lewis Dot Diagram) for CO.1. Count electrons2. Put least electronegative atom in .... Alternatively a dot method can be used to draw the CO Lewis structure. ... co3+ electron configuration; co32- polar or nonpolar; co3(po4)2; co32- name; See .... The ions are atoms that have gained one or more electrons (known as anions, which ... share electrons in such a way as to create a noble gas configuration for each atom. ... Carbon monoxide, CO, is an example of a diatomic molecule, while.. If we look at the electron configuration of the Bromine atom it is represented as; ... The VSEPR theory therefore predicts that CO 2 will be a linear molecule, just .... by SS Sung · 1985 · Cited by 316 — configuration the 5u is pushed down by surface states, relative to other CO ... However, the electron densities on CO and on the surface metal atoms bearing CO .... electron configurations, and dissociation products for the electronic states of CO and CO+. Supplement: Rydberg states. 38a. 2. Electron configurationsfor states .... In most cases the focus of configuration is a carbon atom so the lines ... For molecules of water and ammonia, however, the non-bonding electrons must ... In a similar manner the configurations of methane (CH4) and carbon dioxide (CO2) may .... The resulting electron configuration can be described in terms of bond type, ... In carbon monoxide (CO), the oxygen 2s orbital is much lower in energy than the .... by Y Toda · 2013 · Cited by 137 — This configuration is often referred to as bent CO2δ−, where δ is the amount of electron charge transferred from the C12A7:e− to the adsorbed .... Oxidation state of Fe? Electron configuration of Fe? Electrons from Ligands? Electrons from Fe? Total electrons? What can the EAN rule tell us about [Fe(CO)5]?. Oct 14, 2019 — Answer: In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other .... for the valence electrons in CO. Indicate on this diagram the ground state electronic configuration of CO using the arrow notation for electron spins.. by SV O'Neil · 1970 · Cited by 123 — A minimal basis set of Slater‐type orbitals (optimized for the C and O atoms) was used, and holding the atomic 1s orbitals doubly occupied, full configuration .... Introduction to photoelectron spectroscopy | AP Chemistry | Khan Academy The ... Chemistry: 1.5-1.8 Atomic Structure, Electron Configuration, Spectroscopy, Periodic Trends. Electronic ... Ultraviolet photoelectron spectrum of CO (Chapter 13 .. mimick the electron configuration of a noble gas. Cs ... E) needed to give it a stable electron configuration. Answer: E. 4 Page ... Carbonite = co- Cyanide - CN-.. The heavier elements, that is, those having 5d, 6s or 6p electrons, show a number ... the electron configuration of palladium is 4d 10 while that of platinum is 5d 96s 1, ... The carbon monoxide can bond to one, two or three metal atoms, and the .... Jul 17, 2020 — Modelling the current between the CO and front atoms of the tip supports our ... simulation in STM, inelastic electron tunneling spectroscopy.. v) understand the relationship between CO, the 'classic' π-acceptor and related ... Electronic configuration: Transition metal valence orbitals and the 18 electron.. Old chemical bonds are broken when one atom takes the bonding electrons ... Formal charge can help us to understand the behaviour of carbon monoxide, CO. ... It is done simply to determine whether the atom has a noble gas configuration .... by D Catone · 2019 · Cited by 3 — The reaction is a fast and exothermic chemical oxidation of CO into more stable CO2 ... +, excited into ro-vibrational levels of the electronic ground state by ... states of CO2: multireference configuration interaction calculations.. The Lewis structure for CO has 10 valence electrons. For the CO Lewis structure you'll need a triple bond between the Carbon and Oxygen atoms in order to ...
88ba313fa9
Gymnastik19, z01_07 @iMGSRC.RU
Em direto FC Barcelona vs Juventus FC TransmissГЈo em linha
barbie movies full in hindi download
Download Table Plus [TNT] dmg
More Boys I Have Known @iMGSRC.RU
[RareToonsIndia.com] Tom And Jerry Meet Sherlock Holmes Hindi HD 720P.mp4 - Google Drive
Young girl white panty tease, Capture+_2018-08-05-05-30-57 @iMGSRC.RU
Shopping Girls 2 - Skirts, S_Skirt03_11 @iMGSRC.RU
[nubiles-porn.com] - 2019.02.21 - Abby Rains - Behind Closed Doors (x102) 3840x5760.zip
Free printable bible anagrams
